Calculate The Amount Of Heat
Boil calculate amount temperature ethanol Solved:calculate the heat change at 0^∘ c for each of the following Example problem 3: determining the quantity of heat needed to convert
Solved Calculate the amount of heat energy, q, produced in | Chegg.com
Heat released absorbed calculating Calculating heat released/absorbed Solved calculate the amount of heat required to raise the
Solved calculate the amount of heat needed to boil 156. g of
Heat amount calculating thermodynamics introduction point keySolved calculate the amount of heat needed to melt 196. g of Heat calculate energy amount produced reaction density use ml each chegg show has solved answers solutions question transcribed text problemHeat equation specific energy temperature change mass calorie calories.
Heat thermal converting physicsHeat specific calculate wikihow Raise specific grams transcribedHeat amount calculate melt needed solved.

Heat equation
Heat transfer calculatingHow much heat is released upon converting one mole of steam (18.0 g How to calculate heat capacityHow to calculate heat released and absorbed and heat of reaction part 1.
Heat transfer rate exchanger between two calculating fluidsCalculate molar engines Heat specific calculations temperature lost change gained mass involving presentationCalculate nagwa calculating cooling acts engines.

Heat change
Calculate amount boil needed sure methanolSolved: chapter 1 problem 61e solution Calculating rate of heat transfer between two working fluids of a heatCalculating heat transfer.
Solved calculate the amount of heat needed to boil 37.6 g of[solved] please help b. calculate the amount of heat absorbed by the Steam water convert heat needed quantity chemistrySolved:calculate the heat released when 2.00 l of cl2(g) with a density.

Heat calculate amount raise required substance specific easily change then know if
Solved calculate the amount of heat energy, q, produced inHow to calculate specific heat: 6 steps (with pictures) Specific heat calculating example ppt metal energy if powerpoint presentation calHow to calculate heat capacity.
.


Solved Calculate the amount of heat needed to melt 196. g of | Chegg.com

How To Calculate Heat Capacity - Water as a cooling agent acts

Solved Calculate the amount of heat needed to boil 156. g of | Chegg.com

Heat Equation

Calculating Heat Transfer - YouTube

Solved: Chapter 1 Problem 61E Solution | Chemistry 10th Edition | Chegg.com
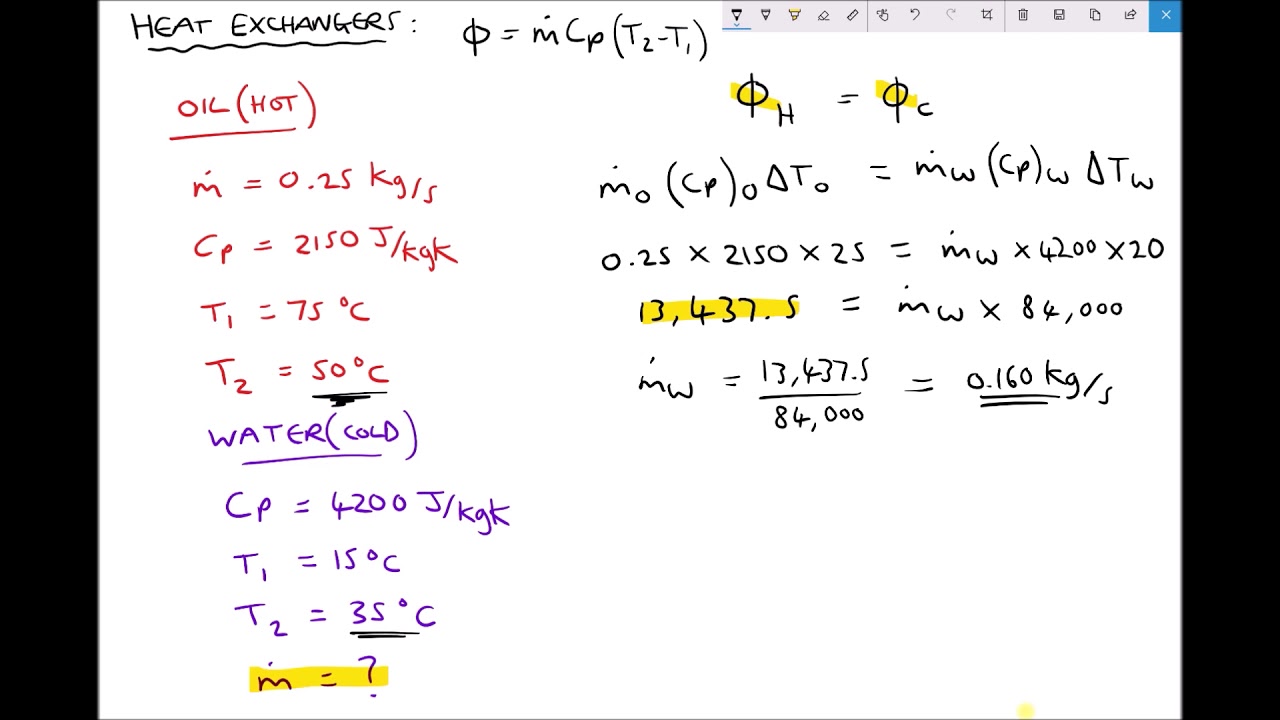
Calculating Rate of Heat Transfer Between Two Working Fluids of a Heat

SOLVED:Calculate the heat change at 0^∘ C for each of the following